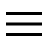Are antibacterial soaps really safe? The FDA has decided to ban 19 substances in antibacterial hand and body soaps for products for sale in the United States.
Don't store avocado like this: it's dangerousAre antibacterial soaps really safe? The FDA has decided to put to the side regarding products for sale in the United States 19 substances present in antibacterial hand and body soaps.
According to the new FDA decision, companies that still use the banned substances in their hand and body hygiene products will have to stop using them within 1 year.
If they do not eliminate them, the products themselves that contain them will be removed from the market. Antibacterial hand and body soaps in the United States apparently contain at least some of these substances, according to Theresa Michele, director of the FDA's Division of Nonprescription Drug Products.
Among the most well-known substances among those that we find in the FDA's viewfinder is the Triclosan, which is used not only in antibacterial soaps but also in municipalities toothpastes And in the intimate cleansers.
As early as 2013, the FDA was thinking of banning Triclosan whose use, according to the American Society Of Microbiology, could increase the risk of infections instead of reducing it.
Read also: ANTIBACTERIAL SOAPS: INCREASE THE RISK OF INFECTIONS DUE TO TRICLOSAN
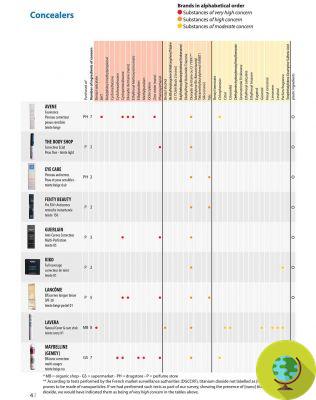
Here the list of substances that the FDA has decided to ban.
The substances reported below with the wording 'Admitted to the EU are currently present in the Inventory of Cosmetic Ingredients of the European Commission which establishes the nomenclature of ingredients used in cosmetic products in Europe. For these ingredients, no restrictions are reported in the European table.
Read also: TRICLOSAN IN DETERGENTS AND COSMETICS SOON BAN IN THE USA?
1) Cloflucarban (antimicrobial, deodorant) - Allowed in the EU
2) Fluorosalan (antimicrobial) - Allowed in the EU
3) Hexachlorophene - Substance considered very toxic in the EU
4) Hexylresorcinol - Here the opinion of EFSA
5) Iodine complex (ammonium ether sulfate and polyoxyethylene sorbitan monolaurate)
6) Iodine complex (phosphate ester of alkylaryloxy polyethylene glycol)
7) Nonylphenoxypoly (ethyleneoxy) ethanoliodine
8) Poloxamer-iodine complex
9) Povidone-iodine (antimicrobial) - Allowed in the EU
10) Undecoylium chloride iodine complex
11) Methylbenzethonium chloride (antimicrobial, deodorn) - Admitted in the EU
12) Phenol (greater than 1.5 percent)
13) Phenol (less than 1.5 percent)
14) Secondary amyltricresols
15) Sodium oxychlorosene
16) Tribromsalan
17) Triclocarban
18) Triclosan (preservative) - Allowed in the EU
19) Triple dye
Consult the European Commission's Cosmetic Ingredient Information Database here.
Marta Albè